I decided to be a little creative when making my most recent lager yeast starter. I am going to be co-brewing an 11 gallon batch of German pils, and this requires a 2 gallon starter using my starter system. It seems like a waste of beer to just dump 2 gallons of starter down the drain, so I decided to make a low gravity beer as my starter instead. I have been meaning to make a berlinerweisse for a while now, and this seemed like the perfect excuse to try it.
I have been contemplating taming wild yeast. What I mean is I want to isolate clonal strains of wild yeast, be it S. cerevisiae or Brettanomyces species. By far the latter is more intriguing, as there are hundreds of S. cerevisiae strains available. To begin taming Brett stains I need two things: a source and a medium to specifically select Brett versus S. cerevisiae. Well, maybe a third thing – patience, as this is going to get tedious.
|
MYPG
|
modified
|
|
0.3% malt extract
|
0.3% malt extract
|
|
0.3% yeast extract
|
0.25% yeast extract
|
|
1% dextrose
|
1% dextrose
|
|
0.5% peptone
|
0.5% peptone
|
|
1.5% agar
|
1.5% agar
|
205-213, 1998) was 195 ppm. Chad Yakobson at brettanomycesproject.com used 1200 ppm, and Jason at sciencebrewer.com used 312 ppm. I decided to try two concentrations to see what grows – 195 ppm and 390 ppm. My stock solution of copper sulfate was 1.95% w/v CuSO4·5H2O dissolved in water, so I added 0 μl, 250μl (195 ppm), or 500 μl (390 ppm) of copper sulfate solution to 25 ml of melted agar per plate.
My control for S. cerevisiae inhibition was Wyeast 3522, and my test yeast mixture was the dregs of Jolly Pumpkin Bam Biere. I chose Wyeast 3522 (WLP550) because that is the primary yeast strain used to ferment Bam Biere.
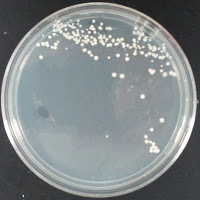 3522 0 ppm CuSO4 |
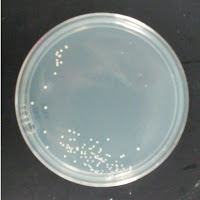 3522 195 ppm CuSO4 |
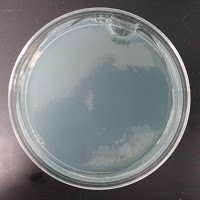 3522 390 ppm CuSO4 |
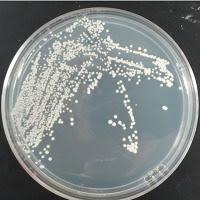 Jolly Pumpkin 0 ppm CuSO4 |
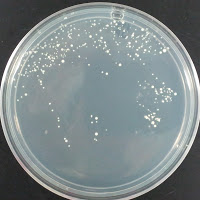 Jolly Pumpkin 195 ppm CuSO4 |
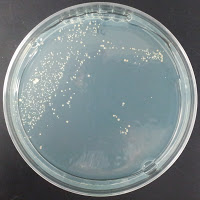 Jolly Pumpkin 390 ppm CuSO4 |
There were no colonies on the 390 ppm plates. I kept the latter plates at 30°C for several more days, and finally something (s) grew on the Jolly Pumpkin plate, but not the Wyeast 3522 plate. I was concerned that what grew out was bacterial in origin, as there is no antibiotic on the plate to suppress bacteria. I picked a couple Jolly Pumpkin colonies, and analyzed them under the microscope, and they were indeed yeast, not bacteria (not shown).
So I decided to try my hand at a partigyle brew, and I figured what better style to do this with than an English brown ale and a mild ale. I’ve been meaning to do this for a few years now, but never sat down to actually calculate a recipe. I sat down with my brown ale recipe (Nutty Brown Dog) and my mild recipe (Muddy Paws Mild), and tried to figure out the commonalities between them so I could formulate a common recipe to brew them both. They both had Maris Otter and chocolate malt, so that part was easy. The crystal malts weren’t too dissimilar – crystal 80 and Special B versus crystal 60 and crystal 120. But the brown ale has special roast and amber malt, while the mild has mild malt and aromatic malt.
|
Nutty Brown Dog
|
Muddy Paws Mild
|
|||
|
9 lb
|
Maris Otter
|
3 lb
|
Maris Otter
|
|
|
8 oz
|
Crystal Malt 80
|
2 lb
|
Mild Malt
|
|
|
8 oz
|
Special Roast
|
1 lb
|
Crystal Malt 60
|
|
|
8 oz
|
Amber Malt
|
8 oz
|
Aromatic Malt
|
|
|
4 oz
|
Chocolate Malt
|
4 oz
|
Crystal Malt 120
|
|
|
3 oz
|
Special B Malt
|
3 oz
|
Chocolate Malt
|
|
|
OG:
|
1.052
|
OG:
|
1.035
|
|
|
SRM:
|
22.8
|
SRM:
|
19.1
|
|
I weighed my options, and in the end I decided to base this recipe off of the mild ale.
To formulate my recipe, I assumed I would obtain 65% of my sugar in the first runnings and 35% in the second runnings. I’ve been analyzing the gravity of my first and second runnings for a few years now, and I typically see 2:1 ratio of the gravity between the first and second runnings. I batch sparge, so I figured this would be predictable of a partigyle. These numbers are similar to those reported by Randy Mosher in Brewing Techniques where he says to expect ~58% of the sugar in the first runnings, and ~42% in the second runnings. I formulated my recipe for an 11 gallon batch assuming 70% efficiency:
9lb Maris Otter
5lb Mild Malt
2lb Crystal Malt 60
8 oz Crystal Malt 120
6 oz Chocolate Malt
1lb Aromatic Malt
OG: 1.042
SRM: 19.6
Given the 65%:35% ratio of first and second runnings, that translates into the following for each:
|
First Runnings (Brown Ale)
|
Second Runnings (Mild)
|
|||
|
5.85 lb
|
Maris Otter
|
3.15 lb
|
Maris Otter
|
|
|
3.25 lb
|
Mild Malt
|
1.75 lb
|
Mild Malt
|
|
|
1.3 lb
|
Crystal Malt 60
|
0.7 lb
|
Crystal Malt 60
|
|
|
5.2 oz
|
Crystal Malt 120
|
2.8 oz
|
Crystal Malt 120
|
|
|
3.9 oz
|
Chocolate Malt
|
2.1 oz
|
Chocolate Malt
|
|
|
0.65 lb
|
Aromatic Malt
|
0.35 lb
|
Aromatic Malt
|
|
|
0.5 oz
|
Challenger 8.3% (60 min)
|
0.5 oz
|
Kent Golding 5.5% (FWH)
|
|
|
0.5 oz
|
Kent Golding 5.5 %(60 min)
|
|||
|
0.5 oz
|
Kent Golding 5.5% (15 min)
|
|||
|
0.5 oz
|
Kent Golding 5.5% (0 min)
|
|||
|
OG:
|
1.055
|
OG:
|
1.029
|
|
|
SRM:
|
25.4
|
SRM:
|
13.7
|
|
|
IBU:
|
27.8
|
IBU:
|
11.4
|
|
I’m always behind on my entries, so here’s a wrap up of the sour cherry ale.
I named it Elysian Cherry Ale, and technically it’s a soured cherry Belgian pale ale.
After primary fermentation (which was quite vigorous), the final gravity was 1.014. I added 1 Lb of cherry juice concentration, which brought it up to 1.022. After a week of secondary fermentation, the gravity was 1.016. I transferred that to a corny keg, and it was force carbonated for a week at about 2.5 vol CO2.
It was then entered into the BUZZ Iron Brewer where it ranked 4th out of fourty-some. The top ten beers then competed in a public tasting where it tied for 2nd. All in all, not too bad.
The flavor was quite unique I think. There is a lot of cherry flavor and aroma, with a slight puckering tartness. It was described as being a little soda-like. Personally I found it enjoyable. I’m definitely going to give it another go, but I’ll do a secondary fermentation with Brett, I think.
I got my wort for the Iron Brewer from Iron Hill on Friday Mar 11. The SG was 1.058, and it was all domestic 2-row.
In short, here’s the recipe:
2.5 oz East Kent Goldings (4.8% AA) – 60 min
Souring:
I want to sour this beer, so what I did was make a sour starter on Tuesday. I made 1 quart of sugar water (SG = 1.032). I added a handful of 2 row to it, and incubated at room temp.
On Friday, I transfered the wort to a cooler, and cooled to about 100F. Then I added the sour starter, and closed it up. Incubated for about 1.5 days, then drained the wort. It was a bit sour, with an aroma reminiscent of sauerkraut. It tastes pretty good.
Partial mash:
I heat 2 quarts of water up to 160F and added the following grains. Incubated at 150F for 30 min.
CaraMunich I 1lb 5oz
Aromatic Malt 11 oz
Drained and sparged with 2 quarts water. Added to wort.
Boiling:
I brought the wort to a boil and added 2.5 oz East Kent Goldings (4.8% AA)
Boiled for 60 minutes, and then chilled
Transfered to fermenter
Topped off to 5.5 gallons with water
The next morning I pitched Wyeast 3522 Belgian Ardennes
Today, I’m going to start a Cherry Melomel. I’ll first ferment 5 gallons of orange blossom honey. Then I’ll rack it to secondary with cherry juice concentrate. Pretty simple – here’s the recipe.
Wow, I’m actually caught up on my notes. Last night I brewed “Nutty Brown Dog”. It’s been over two years since I made that. I’ve been disappointed in the beer for a while. I really liked the first recipe I came up with for it, so I’m going back to that one. The only changes are that I’ll change the crystal 120 to crystal 80, and change up the hops a little bit. I really wanted to go with East Kent Golding hops the whole way through, but there weren’t any available. Here’s the recipe:
Maris Otter Pale Malt 9 lbs
Crystal 80 Malt 0.5 lbs
Special Roast 0.5 lbs
Amber Malt 0.5 lbs
Chocolate Malt 0.25 lbs
0.75 oz First Gold (8% AA) – 60 min
0.25 oz First Gold (8% AA) – 20 min
1 oz Fuggle (4% AA) – 5 min
Starting Gravity: 1.053
Efficiency: 72%
Calculated SRM: 23.5
Mashed at 156F for 1 hour with 1.25 qt water per lb grain. Collected 3 gal wort, batch sparged with 3 gal water at 168F. Boiled for a total of 1 h.
Chilled wort, transferred equal volumes to plastic food-grade buckets, and aerated by shaking for 5 min.
Pitched Wyeast 1098. Started fermenting at 76F.
I had Jolly Pumpkin’s Bam Bier a bit ago, and I loved it. It’s a great low-alcohol beer with tons of flavor, and a nice subtle sourness. I highly recommend buying this if you ever see it. So, I was inspired to try something similar. The style is along the lines of a low gravity saison, but with wild yeast and bacteria in the fermentation. I listened to the “Can You Brew It?” podcast for details on the Bam Bier recipe. Here was my plan: brew a 1.045 saison-ish beer. Ferment 5 gallons with a saison yeast, and then throw some Bam Bier dregs into a half gallon to ferment separately. Then when fermentation is complete, the two would be blended and allow to ferment of a bit longer. Here’s the recipe:
Wildbriar Ale – Brewed July 24, 2010
2-Row Pale Malt 4 lbs
Pilsner Malt 3 lbs
Wheat Malt 1 lbs
Flaked Barley 0.5 lbs
Crystal 80 0.25 lbs
1 oz Spalt (5% AA) – 60 min
0.2 oz Cluster (7% AA) – 45 min
0.5 Hallertauer (3.2% AA) – 15 min
Starting Gravity: 1.045
Efficiency: 77%
Calculated SRM: 3.5
Calculated IBU: 26.85
Mashed at 148F for 1.5 hour with 1.5 qt water per lb grain. Collected 3.2 gal wort, batch sparged with 3 gal water at 168F. Boiled for a total of 1.5 h.
Chilled wort, transferred equal volumes to plastic food-grade buckets, and aerated by shaking for 5 min.
Transferred 0.5 gallon of wort to a 1 gall on jar. Pitched 1 ml of a glycerol stock of dregs from a Jolly Pumpkin Bam Bier.
OK, it’s been a while. Between moving, removing a fallen tree, generally taking care of a one-year-old, and working, it’s been hard to find extra time.
Here’s the questionnaire we presented to the homebrew club a few months ago regarding the English ale yeast strains. In short, everyone could taste differences in all beers. Some strains were more popular than others. It is interesting that 1768 is a very polarizing strain. Some loved it, some hated it. Overall, it was an educational, but labor intensive, experiment. Given the differences between all of these yeasts, I highly recommend comparing different strains when formulating recipes. Comparing eight at a time is not practical (or advisable), but pick two or three and compare the results. Picking one strain every another won’t make or break your recipe, but I guarantee it will make the difference between a good beer and a great beer.
How did the samples actually compare?
Which sample did you enjoy the most?
Which sample did you enjoy the least?
Both 1768 and 1026 seemed to produce strong responses throughout the tasting questionnaire. 1028 and 1098 are significant in that no one indicated that they were least interested in brewing with them.
 Bottled all beers on Sunday (4/25). Added ~1.6 oz dextrose to each, to give us a final CO2 volume of about 1.8. Got an average of 30 bottles from each. The only problem we had was forgetting to add the sugar to the 1028, which we had to pour back into the bottling keg, prime, and rebottle. Other than that, it went pretty well. We got to taste them all side by side, and each was quite different. I’ll post an official tasting once they are done carbonating.
Bottled all beers on Sunday (4/25). Added ~1.6 oz dextrose to each, to give us a final CO2 volume of about 1.8. Got an average of 30 bottles from each. The only problem we had was forgetting to add the sugar to the 1028, which we had to pour back into the bottling keg, prime, and rebottle. Other than that, it went pretty well. We got to taste them all side by side, and each was quite different. I’ll post an official tasting once they are done carbonating.